| Identification |
|---|
| YMDB ID | YMDB00317 |
|---|
| Name | Phosphoribosyl-AMP |
|---|
| Species | Saccharomyces cerevisiae |
|---|
| Strain | Baker's yeast |
|---|
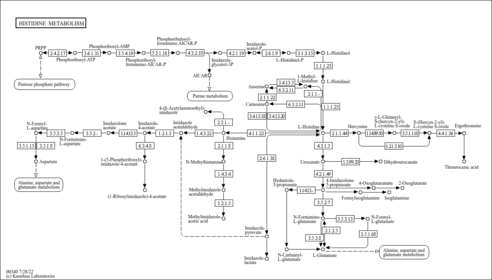
| Description | Phosphoribosyl-AMP, also known as 5-phosphoribosyl-AMP, belongs to the class of organic compounds known as purine ribonucleoside monophosphates. These are nucleotides consisting of a purine base linked to a ribose to which one monophosphate group is attached. Phosphoribosyl-AMP is a very strong basic compound (based on its pKa). Phosphoribosyl-AMP exists in all living species, ranging from bacteria to humans. Within yeast, phosphoribosyl-AMP participates in a number of enzymatic reactions. In particular, phosphoribosyl-AMP can be biosynthesized from 1-(5-phosphoribosyl)-ATP through its interaction with the enzyme histidine biosynthesis trifunctional protein. In addition, phosphoribosyl-AMP can be converted into phosphoribosylformiminoaicar-phosphate; which is mediated by the enzyme histidine biosynthesis trifunctional protein. In yeast, phosphoribosyl-AMP is involved in the metabolic pathway called histidine biosynthesis pathway. |
|---|
| Structure | |
|---|
| Synonyms | - 1-(5-Phosphoribosyl)-AMP
- 5-phosphoribosyl-AMP
- N-(5-phospho-D-ribosyl)-AMP
- N-(5'-phospho-D-ribosyl)-AMP
- N1-(5-phospho-D-ribosyl)-AMP
|
|---|
| CAS number | Not Available |
|---|
| Weight | Average: 559.3157
Monoisotopic: 559.071673493 |
|---|
| InChI Key | RTQMRTSPTLIIHM-ZLRMBSEFSA-N |
|---|
| InChI | InChI=1S/C15H23N5O14P2/c16-12-7-13(18-4-19(12)14-10(23)8(21)5(33-14)1-31-35(25,26)27)20(3-17-7)15-11(24)9(22)6(34-15)2-32-36(28,29)30/h3-6,8-11,14-16,21-24H,1-2H2,(H2,25,26,27)(H2,28,29,30)/b16-12+/t5-,6-,8-,9-,10-,11-,14+,15-/m1/s1 |
|---|
| IUPAC Name | {[(2R,3S,4R,5R)-5-{1-[(2S,3R,4S,5R)-3,4-dihydroxy-5-[(phosphonooxy)methyl]oxolan-2-yl]-6-imino-6,9-dihydro-1H-purin-9-yl}-3,4-dihydroxyoxolan-2-yl]methoxy}phosphonic acid |
|---|
| Traditional IUPAC Name | [(2R,3S,4R,5R)-5-{1-[(2S,3R,4S,5R)-3,4-dihydroxy-5-[(phosphonooxy)methyl]oxolan-2-yl]-6-iminopurin-9-yl}-3,4-dihydroxyoxolan-2-yl]methoxyphosphonic acid |
|---|
| Chemical Formula | C15H23N5O14P2 |
|---|
| SMILES | [H]O[C@@]1([H])[C@@]([H])(O[C@]([H])(C([H])([H])OP(=O)(O[H])O[H])[C@@]1([H])O[H])N1C([H])=NC2=C1N=C([H])N(C2=N[H])[C@@]1([H])O[C@]([H])(C([H])([H])OP(=O)(O[H])O[H])[C@@]([H])(O[H])[C@@]1([H])O[H] |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as purine ribonucleoside monophosphates. These are nucleotides consisting of a purine base linked to a ribose to which one monophosphate group is attached. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Nucleosides, nucleotides, and analogues |
|---|
| Class | Purine nucleotides |
|---|
| Sub Class | Purine ribonucleotides |
|---|
| Direct Parent | Purine ribonucleoside monophosphates |
|---|
| Alternative Parents | |
|---|
| Substituents | - Purine ribonucleoside monophosphate
- Pentose phosphate
- Pentose-5-phosphate
- Glycosyl compound
- N-glycosyl compound
- Monosaccharide phosphate
- Imidazopyrimidine
- Purine
- Monoalkyl phosphate
- Monosaccharide
- N-substituted imidazole
- Organic phosphoric acid derivative
- Phosphoric acid ester
- Pyrimidine
- Alkyl phosphate
- Imidolactam
- Heteroaromatic compound
- Imidazole
- Tetrahydrofuran
- Azole
- Secondary alcohol
- Oxacycle
- Organoheterocyclic compound
- Azacycle
- Organopnictogen compound
- Alcohol
- Organic oxide
- Organic oxygen compound
- Organonitrogen compound
- Organooxygen compound
- Hydrocarbon derivative
- Organic nitrogen compound
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Charge | 0 |
|---|
| Melting point | Not Available |
|---|
| Experimental Properties | | Property | Value | Reference |
|---|
| Water Solubility | Not Available | PhysProp | | LogP | Not Available | PhysProp |
|
|---|
| Predicted Properties | |
|---|
| Biological Properties |
|---|
| Cellular Locations | Not Available |
|---|
| Organoleptic Properties | Not Available |
|---|
| SMPDB Pathways | |
|---|
| KEGG Pathways | |
|---|
| SMPDB Reactions | |
|---|
| KEGG Reactions | Not Available |
|---|
| Concentrations |
|---|
| Intracellular Concentrations | Not Available |
|---|
| Extracellular Concentrations | Not Available |
|---|
| Spectra |
|---|
| Spectra | |
|---|
| References |
|---|
| References: | - UniProt Consortium (2011). "Ongoing and future developments at the Universal Protein Resource." Nucleic Acids Res 39:D214-D219.21051339
|
|---|
| Synthesis Reference: | Not Available |
|---|
| External Links: | |
|---|